How to convert Atmospheric pressure to Pascal (atm-Pa) and Pascal to Atmospheric pressure (Pa-atm). - YouTube

pressure, conversion units into defferent units,atm,bar,torr,psi,Pascal,mmHg, numerical,and examples - YouTube

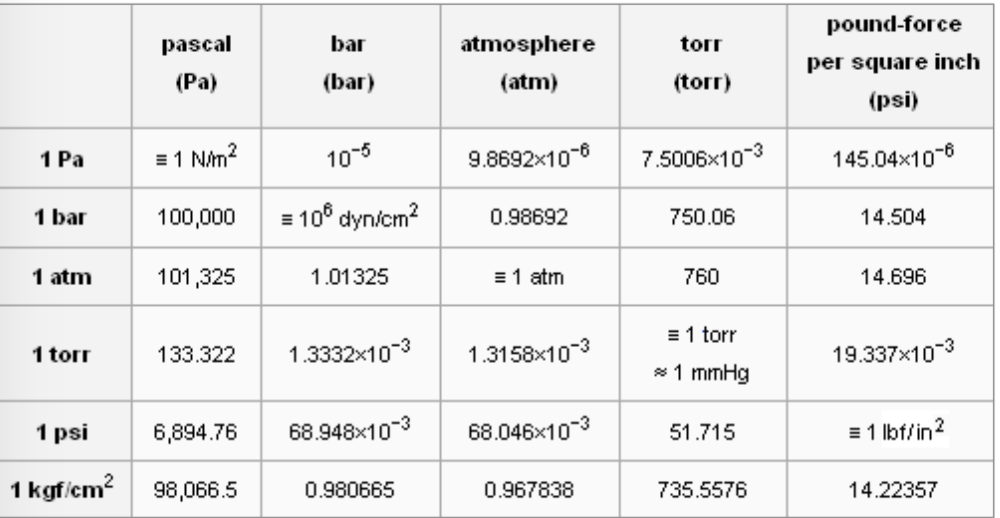
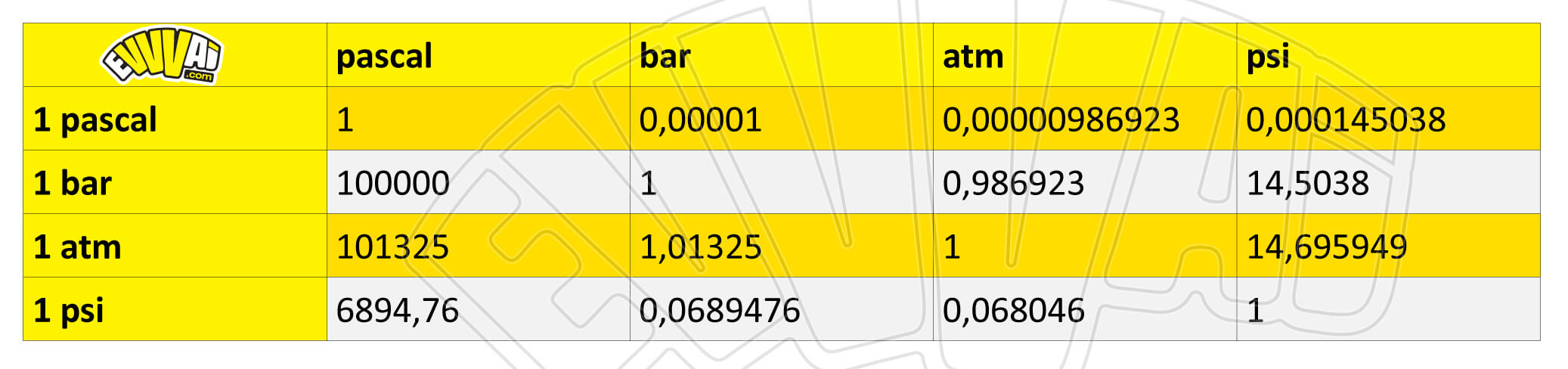
Atmospheres and Conversions: 1.00 atm = x 10 5 Pa = kPa = 760. Torr = 14.7 psi To convert pressure: 1.Turn what you have to atm 2.Multiply. - ppt download

F. Convert the following pressure values into Pascals a. 10 atmosphere c 107000 Nm b. 1 kPa. d. 1 atmosphere

1 atm = 1.013x105 Pa = kPa = 14.7 psi = 760 Torr; 1 m3 = 1000 liters; pabsolute = pgauge + 1 atm; ) Convert Torr absolute to gauge pressure. - ppt download

SOLVED: What is the pressure 40 m underwater? Assume the water surface is at atmospheric pressure (1 atm = 101,300 Pa), and the density of water is p = 1000 kg/m^3. Express

SOLVED: Question 6 0/1p Convert 1.20 atm to Pa 8.44 x 104 Pa 1.18 * 10-5 Pa 1.22 x 105 Pa 101,325 Pa How many Pa are in 1 atm?

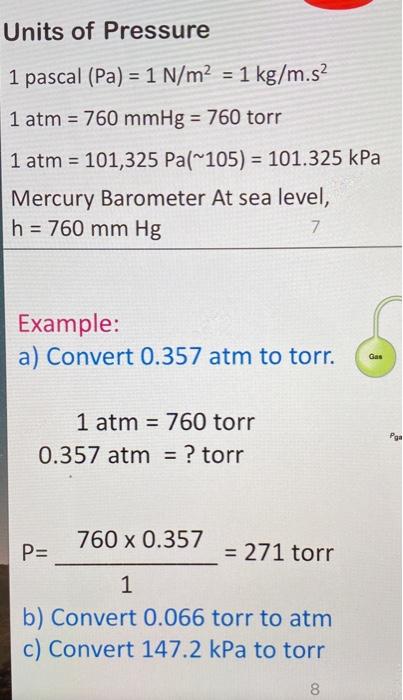
SOLVED: 1 atm = 760 mm Hg 760 torr = 1 atm 1 atm = 101325 Pa 101.3 J = 1 L atm 1 nm = 1 x 10^-9 m 0.08212 M^-1

SOLVED: QUESTION 4 atm is equal to: 760 mm Hg: A 14.7 psi: B: 760 torr. all of the above D 101,325 Pa: E